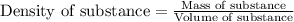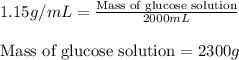Chemistry, 22.06.2019 13:00 taylorpayne525p8qxky
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
1. calculate the approximate enthalpy of the reaction in joules. estimate that 1.0 ml of vinegar has the same thermal mass as 1.0 ml of water. iqnore the thermal mass of th sodium bicarbonate. note: it takes about 4.2 joules () to change 1.0 gram (1.0ml) of water 1.0 c
Answers: 2

Chemistry, 22.06.2019 21:00
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1

Chemistry, 22.06.2019 22:30
The diagram shows the relationship between scientific disciplines.the names of some scientific disciplines have been removed from the boxes. which scientific discipline belongs in the blue box? a.physics b.biology c.chemistry d.metallurgy
Answers: 2

Chemistry, 23.06.2019 01:30
If a particle has z = 25 and 23 electrons, what is its charge?
Answers: 2
You know the right answer?
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?...
Questions

English, 14.06.2021 07:30



Mathematics, 14.06.2021 07:30



Computers and Technology, 14.06.2021 07:30

Mathematics, 14.06.2021 07:30


Social Studies, 14.06.2021 07:40

Chemistry, 14.06.2021 07:40

Chemistry, 14.06.2021 07:40

Mathematics, 14.06.2021 07:40

Biology, 14.06.2021 07:40

Computers and Technology, 14.06.2021 07:40


Mathematics, 14.06.2021 07:40


Chemistry, 14.06.2021 07:40

Biology, 14.06.2021 07:40