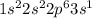Chemistry, 22.06.2019 13:10 kellinvagneur
Which electron configuration represents the electrons in an atom of sodium in the ground state at stp

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 15:20
Water is initially present in a state where its molecules are far apart. during a change of state, its molecules slow down. which change of state has most likely taken place? from a gas to a liquid from a liquid to a gas from a solid to a liquid from a gas to a plasma
Answers: 1

Chemistry, 22.06.2019 20:00
I’m an electrically neutral atomic any element, there are equal numbers of
Answers: 2

Chemistry, 22.06.2019 21:30
If 22.5 of nitrogen at 748 mm hg are compressed to 725 mm hg at constant temperature. what is the new volume?
Answers: 1
You know the right answer?
Which electron configuration represents the electrons in an atom of sodium in the ground state at st...
Questions

Business, 02.03.2021 02:40

History, 02.03.2021 02:40







Health, 02.03.2021 02:40

History, 02.03.2021 02:40





Biology, 02.03.2021 02:40

History, 02.03.2021 02:40


Mathematics, 02.03.2021 02:40

Mathematics, 02.03.2021 02:40