
Chemistry, 05.08.2019 22:20 weridness80
For the titration of a weak acid with a strong base what is the pka of the weak acid if the ph is 6.72 at the equilvalence point (100% t), 9.12 at the end point, and 4.23 at 50% t? answer with one sig fig.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1
You know the right answer?
For the titration of a weak acid with a strong base what is the pka of the weak acid if the ph is 6....
Questions

History, 01.12.2020 20:30

Mathematics, 01.12.2020 20:30


Mathematics, 01.12.2020 20:30

Mathematics, 01.12.2020 20:30

Mathematics, 01.12.2020 20:30


Mathematics, 01.12.2020 20:30



Chemistry, 01.12.2020 20:30





Mathematics, 01.12.2020 20:30

English, 01.12.2020 20:30


Physics, 01.12.2020 20:30

History, 01.12.2020 20:30

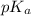 value.
value.![[A^{-}]](/tpl/images/0171/8766/fe74d.png)
![pK_{a} + log \frac{[A^{-}]}{[HA]}](/tpl/images/0171/8766/24c3e.png)
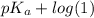 (as [HA] =
(as [HA] = 


