
Chemistry, 10.10.2019 01:00 MyaaaMoney
Gselect the correct statements. i. reduction is the loss of electrons, and oxidation is the gain of electrons ii. reduction is the gain of electrons, and oxidation is the loss of electrons iii. the reactant that is reduced is also called the reducing agent, and the reactant that is oxidized is called the oxidizing agent. iv. the reactant that is oxidized is also called the reducing agent, and the reactant that is reduced is called the oxidizing agent. v. the sum of all the oxidation numbers in a compound is equal to the charge of that compound. a. i and iii only b. ii and iv only c. i, iii, and v only d. ii, iv, and v only e. v only

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1

Chemistry, 23.06.2019 03:30
The molar mass of iron(fe) is 55.8 g/mol. what is the mass in grams of 2.25 moles of iron?
Answers: 1

Chemistry, 23.06.2019 05:30
Stoichiometry- i need with 14 and 15! an explanation would be appreciated!
Answers: 1

You know the right answer?
Gselect the correct statements. i. reduction is the loss of electrons, and oxidation is the gain of...
Questions



Mathematics, 28.12.2020 16:00








Advanced Placement (AP), 28.12.2020 16:10


Computers and Technology, 28.12.2020 16:10

Mathematics, 28.12.2020 16:10

Social Studies, 28.12.2020 16:10


Physics, 28.12.2020 16:10


Mathematics, 28.12.2020 16:10

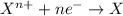
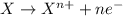
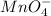 , manganese has +7 oxidation number and oxygen has -2.
, manganese has +7 oxidation number and oxygen has -2.![[=7+(4\times (-2))]=-1](/tpl/images/0305/3031/ac67d.png)


