Chemistry, 30.11.2019 03:31 brooklyn674
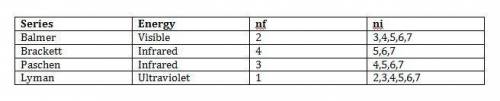
The four sets of lines in the hydrogen emission spectrum are known as balmer, brackett, paschen, and lyman. for each series, assign the energy (infrared, ultraviolet, or visible), the nr value, and all possible n, values up to 7 series balmer brackett paschen lyman energy nf ni visible) infrared ultraviolet 1 2 3 6 7

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:20
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2


Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3
You know the right answer?
The four sets of lines in the hydrogen emission spectrum are known as balmer, brackett, paschen, and...
Questions


History, 06.07.2019 21:00

Biology, 06.07.2019 21:00

History, 06.07.2019 21:00


Biology, 06.07.2019 21:00


History, 06.07.2019 21:00



English, 06.07.2019 21:00









Computers and Technology, 06.07.2019 21:00