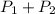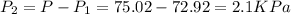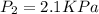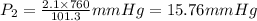Chemistry, 31.03.2020 00:16 rwbrayan8727
A gas is collected by water displacement so that its partial pressure is 72.92 kPa. The total pressure of the gas over water is 75.02 kPa. What is the temperature at which the gas was collected?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:40
If 10.0 ml of the solution on the right are withdrawn from the 100 ml beaker and diluted again in a similar manner, what is the new concentration? m nacl
Answers: 2

Chemistry, 21.06.2019 23:00
Ahypothrticalax type of ceramic material is known to have a density of 2.10 g/cm3 and a unit cell of cubic symmetry with a cell edge length of 0.57 nm. the atomic weights of the a and x elements are 28.5and 30.0 g/mol, respectively. on the basis of this information, which of the following crystal structures is (are) possible for this material: sodium chloride, cesium chloride, or zinc blende
Answers: 1

Chemistry, 22.06.2019 14:30
Which of the following describes a situation where competition between producers exists
Answers: 1

Chemistry, 22.06.2019 14:30
Select all of the statements which are true. electrons are located in shells or orbits around the atom. electrons orbit slowly around the atom. electrons travel in one flat path around the nucleus of an atom. the valence of an atom is determined by the number of electrons in the atom's outermost shell.
Answers: 1
You know the right answer?
A gas is collected by water displacement so that its partial pressure is 72.92 kPa. The total pressu...
Questions

Mathematics, 18.01.2020 13:31

History, 18.01.2020 13:31

English, 18.01.2020 13:31

Mathematics, 18.01.2020 13:31

Biology, 18.01.2020 13:31




Chemistry, 18.01.2020 13:31

Mathematics, 18.01.2020 13:31


English, 18.01.2020 13:31


Mathematics, 18.01.2020 13:31




Mathematics, 18.01.2020 13:31

Mathematics, 18.01.2020 13:31

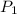 =72.92 KPa
=72.92 KPa