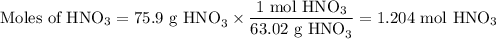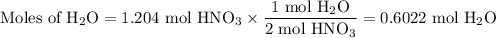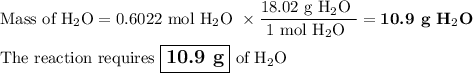Not understanding on how stoichiometry works not sure which side to start on. ex: How many grams of water are required to form 75.9 g of HNO3? Assume that there is excess NO2 present. The molar masses are as follows: H2O = 18.02 g/mol, HNO3 = 63.02 g/mol.
3 NO2(g) + H2O(l) → 2 HNO3(aq) + NO(g)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:10
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2

Chemistry, 22.06.2019 06:30
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2

Chemistry, 22.06.2019 19:50
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3
You know the right answer?
Not understanding on how stoichiometry works not sure which side to start on. ex: How many grams of...
Questions


Chemistry, 04.06.2021 22:00

Social Studies, 04.06.2021 22:00


Mathematics, 04.06.2021 22:00


Mathematics, 04.06.2021 22:00


History, 04.06.2021 22:00






Mathematics, 04.06.2021 22:00

Mathematics, 04.06.2021 22:00


Biology, 04.06.2021 22:00

Mathematics, 04.06.2021 22:00