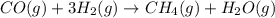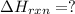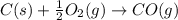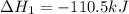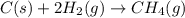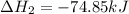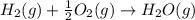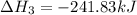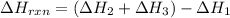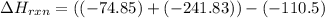Chemistry, 29.06.2020 03:01 priscillavaladez1112
Use Hess's law and the following equations to calculate the ΔHreaction for the reaction CO(g) + 3H2(g) CH4(g) + H2O(g). (Show your work.) (4 points) • C(s) + O2(g) CO(g) ΔH = –110.5 kJ • C(s) + 2H2(g) CH4(g) ΔH = –74.85 kJ • H2(g) + O2(g) H2O(g) ΔH = –241.83 kJ

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:40
Determine the energy released per kilogram of fuel used. given mev per reaction, calculate energy in joules per kilogram of reactants. consider 1 mole of tritium plus 1 mole of deuterium to be a mole of “reactions” (total molar mass = 5 grams).
Answers: 1

Chemistry, 22.06.2019 03:00
Compare the valence electron configuration of the nobles gas elements seen here. what statement is correct?
Answers: 2

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 10:30
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1
You know the right answer?
Use Hess's law and the following equations to calculate the ΔHreaction for the reaction CO(g) + 3H2(...
Questions

Social Studies, 15.04.2021 02:10

Mathematics, 15.04.2021 02:10

Chemistry, 15.04.2021 02:10

English, 15.04.2021 02:10




Mathematics, 15.04.2021 02:10

Mathematics, 15.04.2021 02:10


Mathematics, 15.04.2021 02:10


Social Studies, 15.04.2021 02:10

Mathematics, 15.04.2021 02:10



Spanish, 15.04.2021 02:10



Mathematics, 15.04.2021 02:10