

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 04:30
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3

Chemistry, 23.06.2019 07:00
Scuba divers use tanks of compressed air to them breathe. gases can be compressed because?
Answers: 1
You know the right answer?
The density of whole milk 1.04 g per mL. What is the volume (in quarts) of 18.5 pounds of whole milk...
Questions


English, 10.01.2022 14:00


Computers and Technology, 10.01.2022 14:00

Mathematics, 10.01.2022 14:00



Mathematics, 10.01.2022 14:00

Health, 10.01.2022 14:00

Mathematics, 10.01.2022 14:00

Mathematics, 10.01.2022 14:00


Biology, 10.01.2022 14:00

English, 10.01.2022 14:00

Mathematics, 10.01.2022 14:00


English, 10.01.2022 14:00

Physics, 10.01.2022 14:00

Mathematics, 10.01.2022 14:00

English, 10.01.2022 14:00

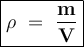 .
.

