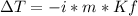Chemistry, 03.06.2021 21:50 versaceblooper
Arrange the following aqueous solutions in order from smallest to greatest freezing point depression: 0.400 m CaCl2, 0.400 m glucose or 0.400 m NaCl, Then determine the freezing point depression for each. For water, Kf = 1.86°C/m.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1

Chemistry, 22.06.2019 19:30
If 16.00g of hydrogen gas reacts with 126.73g of oxygen, how many grams of water are yielded? (both reactants are completely consumed in the reaction.)
Answers: 2

Chemistry, 22.06.2019 19:40
What type of electromagnetic waves does the human eye see as the colors red blue or green a visible light waves b radio waves c infrared waves d microwaves
Answers: 1
You know the right answer?
Arrange the following aqueous solutions in order from smallest to greatest freezing point depression...
Questions




Chemistry, 19.09.2019 11:30


English, 19.09.2019 11:30

Mathematics, 19.09.2019 11:30


Physics, 19.09.2019 11:30


History, 19.09.2019 11:30


Mathematics, 19.09.2019 11:30

Mathematics, 19.09.2019 11:30

Mathematics, 19.09.2019 11:30

Social Studies, 19.09.2019 11:30

Mathematics, 19.09.2019 11:30

Chemistry, 19.09.2019 11:30

History, 19.09.2019 11:30