
Advanced Placement (AP), 25.02.2021 05:40 raquelle66
A student is given a sample of CuSO4(s)
that contains a solid impurity that is soluble and colorless. The student wants to determine the amount of CuSO4
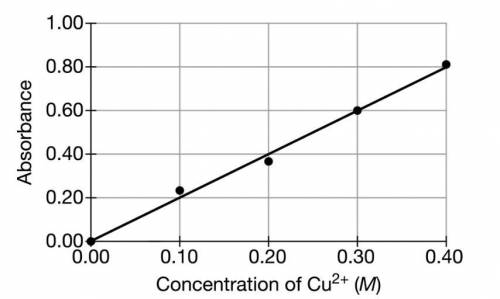
in the sample and decides to use a spectrophotometer. First, the student prepares a calibration graph by measuring the absorbances of CuSO4(aq)
solutions of known concentrations. The graph is shown below.
(a) The student dissolves the entire impure sample of CuSO4(s)
in enough distilled water to make 100.mL
of solution. Then the student measures the absorbance of the solution and observes that it is 0.30
. Determine the concentration of CuSO4(aq)
in the solution.
(b) Calculate the number of moles of CuSO4
that were in the impure sample of CuSO4(s)
.
(c) In addition to the number of moles of CuSO4
calculated in part (b)
, what other quantity must be measured in order to calculate the mass percentage of CuSO4
in the impure sample of CuSO4(s)
?
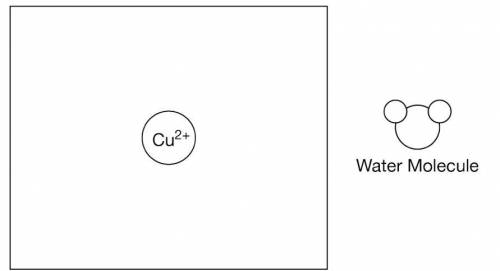
d) A particulate drawing of a single Cu2+
ion is shown in the following box. Draw two H2O
molecules in the box to show the most likely orientation of H2O(l)
molecules around the Cu2+
ion.

Answers: 3


Another question on Advanced Placement (AP)

Advanced Placement (AP), 23.06.2019 19:30
Briefly explain one difference in the practice of realpolitik by louis napoleon and otto von bismarck.
Answers: 1

Advanced Placement (AP), 24.06.2019 07:40
Compare and contrast the environmental consequences of reliance on coal with reliance on petroleum.
Answers: 2


Advanced Placement (AP), 28.06.2019 16:30
Whats 97 x 21.2344557 divided by ………… and tons of ! ok dont answer that ^ i found a son and hes missing now son come back
Answers: 1
You know the right answer?
A student is given a sample of CuSO4(s)
that contains a solid impurity that is soluble and colorles...
Questions

Mathematics, 05.10.2019 11:30



Mathematics, 05.10.2019 11:30


Mathematics, 05.10.2019 11:30




History, 05.10.2019 11:30





Geography, 05.10.2019 11:30


Mathematics, 05.10.2019 11:30

History, 05.10.2019 11:30




