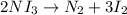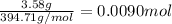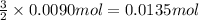Chemistry, 21.07.2019 06:00 itzmelanie1
The chemical equation below shows the decomposition of nitrogen triiodide (ni3) into nitrogen (n2) and iodine (i2). 2ni3 mc030-1.jpg n2 + 3i2 the molar mass of i2 is 253.80 g/mol, and the molar mass of ni3 is 394.71 g/mol. how many moles of i2 will form 3.58 g of ni3?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:00
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1


Chemistry, 22.06.2019 18:30
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2

Chemistry, 23.06.2019 02:00
Alice did an experiment to find the relationship between the angle at which a ray of light strikes a mirror and the angle at which the mirror reflects the light. she placed a ray box in front of a mirror. she changed the angle at which the light from the ray box struck the mirror and noted the corresponding angle at which the mirror reflected the light. which of the following is the dependent variable in this experiment? the mirror used to reflect the light the ray box used as the source of light angle at which the light from the ray box strikes the mirror angle at which the mirror reflects the light from the ray box
Answers: 2
You know the right answer?
The chemical equation below shows the decomposition of nitrogen triiodide (ni3) into nitrogen (n2) a...
Questions


Health, 22.10.2019 11:00

Mathematics, 22.10.2019 11:00

Arts, 22.10.2019 11:00


Mathematics, 22.10.2019 11:00



Mathematics, 22.10.2019 11:00


Arts, 22.10.2019 11:00






Mathematics, 22.10.2019 11:00


Health, 22.10.2019 11:00