Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:20
Concerning the 10.0 ml of 0.50 m nacl to 100 ml of solution: when a solution is diluted, does it change the number of moles dissolved?
Answers: 3

Chemistry, 22.06.2019 01:30
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 03:30
Calculate the molar mass of aluminum oxide (al2o3). express your answer to four significant figures.
Answers: 1

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3
You know the right answer?
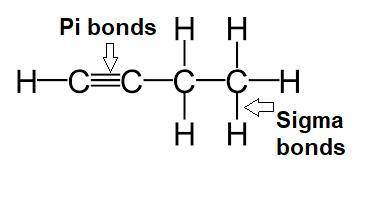
Draw the lewis structure for the molecule ch3ch2cch. how many sigma and pi bonds does it contain?...
Questions

Mathematics, 17.11.2019 22:31


Mathematics, 17.11.2019 22:31

Mathematics, 17.11.2019 22:31


English, 17.11.2019 22:31

Mathematics, 17.11.2019 22:31

Biology, 17.11.2019 22:31




Mathematics, 17.11.2019 22:31

Biology, 17.11.2019 22:31

Mathematics, 17.11.2019 22:31

Physics, 17.11.2019 22:31

Mathematics, 17.11.2019 22:31

Biology, 17.11.2019 22:31

Social Studies, 17.11.2019 22:31


Mathematics, 17.11.2019 22:31