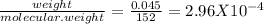Chemistry, 20.07.2019 15:30 mariehayes099
Vanillin, c8h8o3 (m = 152 g/mol), is the molecule responsible for the vanilla flavor in food. how many oxygen atoms are present in a 45.0 mg sample of vanillin?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 19:00
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1

Chemistry, 22.06.2019 21:30
What is another way to determine mass times acceleration?
Answers: 1

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 4.5 × 106 m/s. show your work. note: h= plank's constant (6.62607 x 10-34 j s)
Answers: 1

Chemistry, 23.06.2019 04:31
How does a sample of helium at 15 degree celsius compare to a sample of helium at 215 k? a) the helium at 15 degrees celsius has a higher average kinetic energy that the sample at 215 k. b) the helium at 15 degrees celsius has lower nuclear energy that the sample at 215 k. c) the helium at 15 degrees celsius has slower- moving atoms that the sample at 215 k. d) the helium at 15 degrees celsius has smaller atoms than the sample at 215 k.
Answers: 1
You know the right answer?
Vanillin, c8h8o3 (m = 152 g/mol), is the molecule responsible for the vanilla flavor in food. how ma...
Questions





Social Studies, 10.12.2021 20:20


Mathematics, 10.12.2021 20:20


English, 10.12.2021 20:20





Mathematics, 10.12.2021 20:20

Computers and Technology, 10.12.2021 20:20


Advanced Placement (AP), 10.12.2021 20:20

History, 10.12.2021 20:20

Mathematics, 10.12.2021 20:20

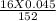 = 0.00473 g
= 0.00473 g