
1. a scientist studies the reaction 2no2(g) 2no(g) + o2(g). she performs three experiments using different concentrations of no2 and measures the initial reaction rate. experiment : [no2] (mol/l) : initial rate ((mol/l)/s) 1 : 0.1 : 0.006 2 : 0.3 : 0.054 3 : 0.5 : 0.150 a. what is the ratio of the concentrations between trials 1 and 2? (2 points) b. what is the ratio of the initial reaction rates between trials 1 and 2? (2 points) c. what is the exponent for [no2] in the rate law? (2 points) d. write the rate law. (2 points) e. solve for the value of k. (2 points) f. what is the overall reaction order? (2 points)

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 22.06.2019 23:30
Why do oxygen have a strong attractive force for electrons
Answers: 2

You know the right answer?
1. a scientist studies the reaction 2no2(g) 2no(g) + o2(g). she performs three experiments using dif...
Questions

Arts, 23.10.2020 07:01

Mathematics, 23.10.2020 07:01


Mathematics, 23.10.2020 07:01

Mathematics, 23.10.2020 07:01


Social Studies, 23.10.2020 07:01

English, 23.10.2020 07:01

English, 23.10.2020 07:01


English, 23.10.2020 07:01

Mathematics, 23.10.2020 07:01

History, 23.10.2020 07:01


Mathematics, 23.10.2020 07:01



Mathematics, 23.10.2020 07:01


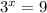 , which gives x = 2, and the exponent in the rate law is 2.
, which gives x = 2, and the exponent in the rate law is 2.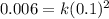 , which yields k = 0.6 L/mol-s.
, which yields k = 0.6 L/mol-s.

