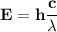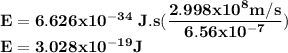Chemistry, 18.07.2019 09:00 hamada11617
One of the wavelengths of light emitted by hydrogen atoms is 6.56 x 10-7 m. calculate the energy.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 22.06.2019 18:30
Two people each hold the end of a rope and create waves by moving their arms up and down. this wave is best classified as a transverse wave because a) both the rope particles and the wave are moving in the same direction. b) the wave is moving up and down as the particles of the rope move horizontally. c) the wave is moving horizontally as the particles of the rope move up and down. eliminate d) the wave is moving in a parallel direction with the motion of the person's arms.
Answers: 3

Chemistry, 22.06.2019 22:20
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2

You know the right answer?
One of the wavelengths of light emitted by hydrogen atoms is 6.56 x 10-7 m. calculate the energy....
Questions

English, 26.06.2019 12:30



Health, 26.06.2019 12:30


Mathematics, 26.06.2019 12:30

Health, 26.06.2019 12:30

History, 26.06.2019 12:30

Biology, 26.06.2019 12:30

Mathematics, 26.06.2019 12:30

Health, 26.06.2019 12:30




Mathematics, 26.06.2019 12:30


Biology, 26.06.2019 12:30