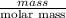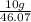Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
Butadiene undergoes a reaction at a certain temperature in the gas phase as follows: 2c4h6(g) --> c8h12(g) the following data were collected for this reaction: time (min) [c4h6] (m) 0 0.36 15 0.30 30 0.25 48 0.19 75 0. determine the order of the reaction and the rate constant. 1st order and k = 4.3x10 -4 s-1 1st order and k = 2.3x10-4 s-1 2nd order and k = 4.3x10-4 s-1 2nd order and k = 2.3x10-4 s-1 zero and k = 4.3x10-4 s-1
Answers: 3

Chemistry, 23.06.2019 04:00
What changes occur in the reaction indicated by the equation? check all that apply. the hydrogen nucleus loses protons. the oxygen nucleus gains protons. the bond in h2 is broken, and new bonds are formed between hydrogen and oxygen atoms. each electron associated with a hydrogen atom is shared with an oxygen atom.
Answers: 3

Chemistry, 23.06.2019 16:10
What type of reaction is shown below? check all that apply. agno3(aq) + nacl(aq) → nano3(aq) + agcl(s) i synthesis decomposition combustion i single replacement double replacement done
Answers: 2

Chemistry, 23.06.2019 22:40
For the following reaction, 3.70 grams of oxygen gas are mixed with excess carbon monoxide . the reaction yields 8.25 grams of carbon dioxide . carbon monoxide(g) + oxygen(g) carbon dioxide(g) what is the ideal yield of carbon dioxide? grams what is the percent yield for this reaction? %
Answers: 1
You know the right answer?
C2h6o how many moles of ethanol are present in a 10.0 g sample of ethanol...
Questions



Chemistry, 25.09.2020 05:01

Mathematics, 25.09.2020 05:01

History, 25.09.2020 05:01

Mathematics, 25.09.2020 05:01

English, 25.09.2020 05:01


Mathematics, 25.09.2020 05:01

Computers and Technology, 25.09.2020 05:01


Physics, 25.09.2020 05:01

Mathematics, 25.09.2020 05:01

Mathematics, 25.09.2020 05:01

Mathematics, 25.09.2020 05:01


History, 25.09.2020 05:01

Mathematics, 25.09.2020 05:01