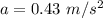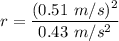Chemistry, 16.07.2019 10:30 Karumbafaith
Asquirrel runs at a steady rate of 0.51 m/s in a circular path around a tree. if the squirrel's centripetal acceleration is 0.43 m/s2, what is the radius of the circle? a.0.60 m b.1.2 m c.0.36 m d.0.84 m

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:30
The reaction q+r2=r2q is found to be first order in r2 and
Answers: 1

Chemistry, 22.06.2019 12:00
Ageochemist examines a piece of metal that he found in the soil. he performs tests to identify the metal from its density, electrical conductivity, and melting point. which statement best describes his investigation? a. he is determining physical properties that are sufficient to identify the metal.b. he is determining chemical properties that are sufficient to identify the metal.c. he is determining physical properties that are insufficient to identify the metal.d. he is determining chemical properties that are insufficient to identify the metal.
Answers: 3

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 23:00
What is the average rate of the reaction between 10 and 20 s?
Answers: 1
You know the right answer?
Asquirrel runs at a steady rate of 0.51 m/s in a circular path around a tree. if the squirrel's cent...
Questions


Mathematics, 26.10.2020 21:40


Mathematics, 26.10.2020 21:40

Arts, 26.10.2020 21:40




Mathematics, 26.10.2020 21:40



History, 26.10.2020 21:40

Social Studies, 26.10.2020 21:40



Business, 26.10.2020 21:40

Social Studies, 26.10.2020 21:40

Chemistry, 26.10.2020 21:40


Mathematics, 26.10.2020 21:40