
Chemistry, 15.07.2019 17:00 imoreno8412
Calculate the enthalpy change for the thermite reaction: 2al(s)+fe2o3(s)→2fe(s)+al2o3(s), δh∘rxn=−850 kj when 12.0 mol of al undergoes the reaction with a stoichiometrically equivalent amount of fe2o3.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 11:30
Compare and contrast refraction of light and sound will give brainliest
Answers: 1

Chemistry, 22.06.2019 12:00
Why are people not able to skip a dive to the deepest part of the ocean
Answers: 1

Chemistry, 22.06.2019 21:30
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3
You know the right answer?
Calculate the enthalpy change for the thermite reaction: 2al(s)+fe2o3(s)→2fe(s)+al2o3(s), δh∘rxn=−8...
Questions

Physics, 18.08.2021 06:20


Physics, 18.08.2021 06:20

Geography, 18.08.2021 06:20

Mathematics, 18.08.2021 06:20



Mathematics, 18.08.2021 06:20



Business, 18.08.2021 06:20



Social Studies, 18.08.2021 06:20


Mathematics, 18.08.2021 06:20


Mathematics, 18.08.2021 06:20

English, 18.08.2021 06:20

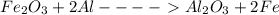
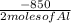 = -5100 KJ
= -5100 KJ

