

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

Chemistry, 22.06.2019 18:00
Chlorophyll a had the molecular formula c55h72mgn4o5 how many atoms are in this molecule
Answers: 2
You know the right answer?
Aseries stack of alkaline cells produces 4.524 kw of power. if each cell contributes 14.5 w, how man...
Questions






Computers and Technology, 12.08.2020 08:01



Mathematics, 12.08.2020 08:01

Mathematics, 12.08.2020 08:01

History, 12.08.2020 08:01



Physics, 12.08.2020 08:01

Mathematics, 12.08.2020 08:01


Mathematics, 12.08.2020 08:01

Mathematics, 12.08.2020 08:01


Mathematics, 12.08.2020 08:01

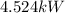 x
x 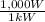 = 4,524W.
= 4,524W. 

