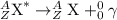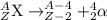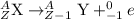After a radioactive atom decays, it is the same element that it was before with no measurable change in mass. which kind of decay has occurred, and how do you know? a) alpha decay because alpha particles have no mass b) beta decay because this kind of decay cannot change one element into another c) alpha decay because it creates a new isotope of the same element d) gamma decay because photons have no mass

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Which function is performed by earths atmosphere? a. ultraviolet rays are prevented from reaching the ozone layer. b. earths temperature is raised and moderated by trapping in heat c. charged particles from the sun are prevented from reaching earth. d. magnetic charges from space are prevented from reaching earths surface.
Answers: 2

Chemistry, 22.06.2019 04:00
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution?
Answers: 2

Chemistry, 22.06.2019 04:40
In which environment would primary succession occur? a forest with a few remaining trees after a recent wildfire an area of exposed rock after a glacier melts away beach that is exposed to the air at low tide an abandoned baseball field in a small town
Answers: 1

Chemistry, 22.06.2019 14:50
How are evaporation and sublimation similar? a both involve the formation of a gas. b both release energy to the surroundings. c both take place throughout a solid. d both take place at the surface of a liquid.
Answers: 1
You know the right answer?
After a radioactive atom decays, it is the same element that it was before with no measurable change...
Questions





Biology, 02.08.2019 19:30


Biology, 02.08.2019 19:30

Spanish, 02.08.2019 19:30


History, 02.08.2019 19:30

Biology, 02.08.2019 19:30


Biology, 02.08.2019 19:30

Mathematics, 02.08.2019 19:30



Business, 02.08.2019 19:30

Mathematics, 02.08.2019 19:30