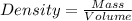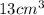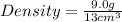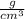Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3


Chemistry, 22.06.2019 17:00
Complete each row of the table below by filling in the missing prefix or missing exponent.
Answers: 1

Chemistry, 23.06.2019 00:30
Balance the following reaction. as2s3 + 9o2 → 2as2o3 + so2
Answers: 2
You know the right answer?
What is the density of a 9.0 gram object that displaces 13 cm3 of water?...
Questions

History, 02.02.2020 15:55



Mathematics, 02.02.2020 15:55


History, 02.02.2020 15:55

History, 02.02.2020 15:55


Biology, 02.02.2020 15:55


English, 02.02.2020 15:55

Mathematics, 02.02.2020 15:55



English, 02.02.2020 15:55

Mathematics, 02.02.2020 15:55