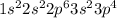Which of these items best describes the arrangement of electrons in an atom of sulfur? a. eight electrons in its first energy level; eight valence electrons in its outermost shell b. two electrons in its first energy level; two electrons in its second energy level; eight electrons in its third energy level; four valence electrons in its outermost energy level c. two valence electrons in its first energy level; eight electrons in its second energy level; six electrons in its outermost energy level d. two electrons in its first energy level; eight electrons in its second energy level; six valence electrons in its outermost energy level

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
For ai it's atomic number is 13 and it's mass number is 27 how many neutrons does it have
Answers: 1

Chemistry, 22.06.2019 13:00
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3

Chemistry, 22.06.2019 18:00
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1
You know the right answer?
Which of these items best describes the arrangement of electrons in an atom of sulfur? a. eight ele...
Questions






English, 20.09.2019 05:30

Social Studies, 20.09.2019 05:30

Chemistry, 20.09.2019 05:30

Social Studies, 20.09.2019 05:30


Mathematics, 20.09.2019 05:30





Mathematics, 20.09.2019 05:30


Biology, 20.09.2019 05:30

History, 20.09.2019 05:30