Answers: 2


Another question on Chemistry

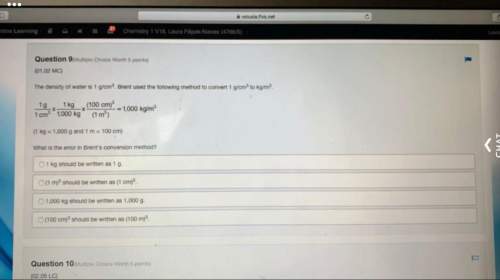
Chemistry, 22.06.2019 18:10
Given the following at 25c calculate delta hf for hcn (g) at 25c. 2nh3 (g) +3o2 (g) + 2ch4 (g) > 2hcn (g) + 6h2o (g) delta h rxn= -870.8 kj. delta hf=-80.3 kj/mol for nh3 (g), -74.6 kj/mol for ch4, and -241.8 kj/mol for h2o (g)
Answers: 1

Chemistry, 22.06.2019 19:30
What is the common name for the compound shown here? enter the common name of the compound shown?
Answers: 2

Chemistry, 22.06.2019 20:10
What would happen to a volleyball left outside in the winter? o o o o a. it would expand. b. it would lose air. c. it would shrink. d. it would explode.
Answers: 2

You know the right answer?
As a candle burns, the chemical energy stored in the wax is transformed into (1) heat and light (2)...
Questions




English, 23.06.2021 14:00

Mathematics, 23.06.2021 14:00

Mathematics, 23.06.2021 14:00

Spanish, 23.06.2021 14:00

Physics, 23.06.2021 14:00


Chemistry, 23.06.2021 14:00


Physics, 23.06.2021 14:00


Mathematics, 23.06.2021 14:00

Mathematics, 23.06.2021 14:00

Mathematics, 23.06.2021 14:00

English, 23.06.2021 14:00


Mathematics, 23.06.2021 14:00