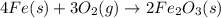Another real-world example of a change is rust. rust is iron oxide (fe2o3), which results when iron is exposed to oxygen in the air. what are some indicators of rusting? check all that apply. production of light color change formation of a solid formation of a gas

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:00
Asample of gas occupies 17 ml at –112°c. what volume does the sample occupy at 70°c a. 10.6 ml b. 27 ml c. 36 ml d. 8.0 ml you
Answers: 1

Chemistry, 22.06.2019 00:30
13. calculate the initial concentration (before precipitation) of carbonate ions after the addition of each 0.05 ml of solution b to the 1.00 l beaker of solution a. divide the work among group members and write the answers in the table in model 3. assume the volume change as solution b is added is negligible. 14. notice the initial concentrations of zn2+ - and cu2+ in the table in model 3. a. explain how these were obtained from the data in model 2. b. as solution b is added and precipitates form, do these initial concentrations change? 15. use the data in model 2 to indicate the presence of precipitate (either znco3 or cuco3) after each 0.05 ml addition of solution b in model 3. 16. use the initial concentrations of carbonate ions and zinc ions to calculate the reaction quotient, qsp for the zinc carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3. 17. use the initial concentrations of carbonate ion and copper(ii) ions to calculate the qsp for the copper(ii) carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3.
Answers: 3

Chemistry, 22.06.2019 06:00
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1
You know the right answer?
Another real-world example of a change is rust. rust is iron oxide (fe2o3), which results when iron...
Questions


Mathematics, 02.02.2021 23:40


Law, 02.02.2021 23:40

Mathematics, 02.02.2021 23:40




Mathematics, 02.02.2021 23:40



Mathematics, 02.02.2021 23:40

Computers and Technology, 02.02.2021 23:40

History, 02.02.2021 23:40

Mathematics, 02.02.2021 23:40


Mathematics, 02.02.2021 23:40

Mathematics, 02.02.2021 23:40


History, 02.02.2021 23:40

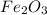 ) which is formed when iron comes in contact with oxygen. Equation for rusting follows:
) which is formed when iron comes in contact with oxygen. Equation for rusting follows: