

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:50
An aqueous solution of potassium hydroxide is standardized by titration with a 0.194 m solution of hydrobromic acid. if 25.2 ml of base are required to neutralize 24.2 ml of the acid, what is the molarity of the potassium hydroxide solution? m potassium hydroxide
Answers: 2


Chemistry, 23.06.2019 05:40
Why is it incorrect to balance a chemical equation by changing the subscripts? explain.
Answers: 2

Chemistry, 23.06.2019 08:00
Pl what kind of reaction is this? nahco3 + h2o → co2 + naoh + h2o -composition -decomposition -single replacement -double replacement im leaning more toward single replacement. if im wrong can you explain whyy?
Answers: 2
You know the right answer?
The ion p3− has protons and electrons. enter your answers as integers separated by a comma.'...
Questions








English, 22.01.2022 01:20



Biology, 22.01.2022 01:20

Social Studies, 22.01.2022 01:20

English, 22.01.2022 01:30







Mathematics, 22.01.2022 01:30

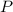 is for phosphorus having atomic number 15. That means it has
is for phosphorus having atomic number 15. That means it has 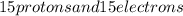 in its neutral state.
in its neutral state.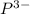 has a -3 charge that means it has gained 3 electrons so, the number of electrons in the
has a -3 charge that means it has gained 3 electrons so, the number of electrons in the 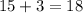 . The number of protons will remain same that is the number of protons = 15.
. The number of protons will remain same that is the number of protons = 15.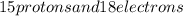 that gives
that gives 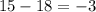 charge on the
charge on the 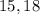 .
. 

