Chemistry, 12.07.2019 12:30 markmlg122
An unknown compound with a molar mass of 155.06 g/mol consists of 46.47% c, 7.80% h, and 45.72% cl. find the molecular formula for the compound. c6h12cl2 chcl c9h18cl3 c6h12cl

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3

Chemistry, 22.06.2019 22:30
Which of the following molecules is polar? c3h7oh c2h5cooh
Answers: 1


Chemistry, 23.06.2019 10:00
Abike ride event is 30 miles. a first aid tent is put at the 3/4 mark of the course. how many miles from the starting point is the first aid tent?
Answers: 1
You know the right answer?
An unknown compound with a molar mass of 155.06 g/mol consists of 46.47% c, 7.80% h, and 45.72% cl....
Questions


Business, 13.03.2021 16:00

English, 13.03.2021 16:10

Geography, 13.03.2021 16:10

Mathematics, 13.03.2021 16:10



Mathematics, 13.03.2021 16:10

History, 13.03.2021 16:10


Mathematics, 13.03.2021 21:30

Arts, 13.03.2021 21:30






Mathematics, 13.03.2021 21:30


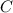 :
: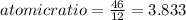
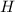 :
: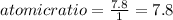
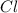 :
: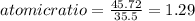
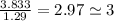
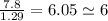
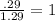
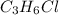 .
.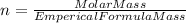
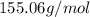

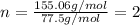
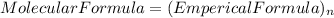 so, the molecular formula is:
so, the molecular formula is: