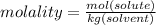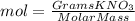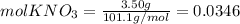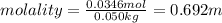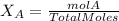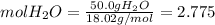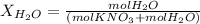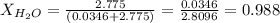Chemistry, 12.07.2019 12:00 Pizzapegasus1
Calculate the molality and mole fraction of water, respectively, of a solution that is made by dissolving 3.50 g of potassium nitrate in 50.0 g of water. the final volume of the solution is 56.0 ml.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 14:00
Write skeleton equations for the following reactions c. aluminum(s)+copper(i) chloride(aq) > aluminum chloride(aq)+copper(s)
Answers: 1

Chemistry, 22.06.2019 02:30
You have a sample of a gas that occupies a volume of 17ml at -111 degrees celsius. what volume does the sample occupy at 88 degrees celsius? show all work asap
Answers: 3

Chemistry, 22.06.2019 15:00
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

Chemistry, 23.06.2019 10:30
Identify the limiting reactant when 9.65-g h2so4 reacts with 6.10-g of naoh.the equation is h2s04 + 2naoh = 2h2o + na2so4• what is the theoretical yield of na2so4, in grams? • how much of the excess reagent will remain after the reaction has been completed? • if 10.5-g of na2so4 are actually recovered experimentally, what is the percent yield?
Answers: 3
You know the right answer?
Calculate the molality and mole fraction of water, respectively, of a solution that is made by disso...
Questions




Biology, 16.07.2019 20:10



History, 16.07.2019 20:10



Mathematics, 16.07.2019 20:10





History, 16.07.2019 20:10




Mathematics, 16.07.2019 20:20