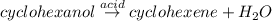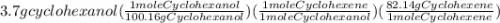Chemistry, 12.07.2019 05:30 ayoismeisjjjjuan
Areaction was performed in which 3.7 g of cyclohexanol was reacted with an acid catalyst to obtain 2.6 g of cyclohexene. calculate the theoretical yield for this reaction.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:20
In a reaction equation, where are the products located? a.) above the arrow b.) to the right of the arrow c.) to the left of the arrow d.) below the arrow
Answers: 2

Chemistry, 22.06.2019 17:50
Cryolite, na3alf6(s), an ore used in the production of aluminum, can be synthesized using aluminum oxide. start this question by first balance the chemical equation.1.) balance the equation: - alo3(s)+naoh(l)+hf(> na3alf6+h2o(g). 2.) if 17.5 kilograms of al2o3(s), 51.4 kilograms of naoh(l), and 51.4 kilograms of hf(g) react completely, how many kilograms of cryolite will be produced? 3.)which reactants will be in excess, (al2o3, naoh, or hf) 4.)what is the total mass of the excess reactants left over after the reaction is complete in kg?
Answers: 2

Chemistry, 22.06.2019 19:00
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1

Chemistry, 22.06.2019 20:30
Draw a line graph showing the relationship between temperature in kelvin as a function of kinetic energy.
Answers: 3
You know the right answer?
Areaction was performed in which 3.7 g of cyclohexanol was reacted with an acid catalyst to obtain 2...
Questions

History, 15.09.2020 01:01

History, 15.09.2020 01:01

History, 15.09.2020 01:01

Biology, 15.09.2020 01:01

English, 15.09.2020 01:01

English, 15.09.2020 01:01

Arts, 15.09.2020 01:01

English, 15.09.2020 01:01

English, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

English, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Spanish, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01