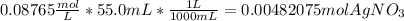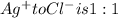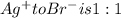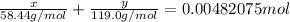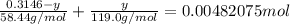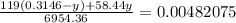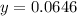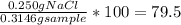Chemistry, 11.07.2019 19:00 alliwkatring
A0.3146-g sample of a mixture of nacl(s) and kbr(s) was dissolved in water. the resulting solution required 55.00 ml of 0.08765 m agno3(aq) to precipitate the cl–(aq) and br–(aq) as agcl(s) and agbr(s). calculate the mass percentage of nacl(s) in the mixture.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:00
Nickel crystallizes in the face-centered cubic (fcc) lattice. the density of the metal is 8902 kg/m3. calculate the radius of a nickel atom.
Answers: 1


Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 22.06.2019 16:00
Which process transfers heat from inside earth to its surface? convection currents in mantle pulling away of tectonic plates drawing in of tectonic plates convection currents in crust
Answers: 1
You know the right answer?
A0.3146-g sample of a mixture of nacl(s) and kbr(s) was dissolved in water. the resulting solution r...
Questions

English, 03.05.2021 15:10







History, 03.05.2021 15:10

Mathematics, 03.05.2021 15:10





History, 03.05.2021 15:10

Biology, 03.05.2021 15:10







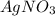 =
=