
Chemistry, 11.07.2019 07:00 emhunter101
Three samples of the same metal are weighed and their masses are found to be 44.40 g, 40.58 g, and 38.35 g. the corresponding volumes are 4.8 ml, 4.7 ml, and 4.2 ml, respectively. determine the density, d, of the metal and calculate the standard deviation of the density.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:50
How many grams of oxygen gas are contained in a 15 l sample at 1.02 atm and 28°c? show your work.
Answers: 1

Chemistry, 22.06.2019 18:00
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2


You know the right answer?
Three samples of the same metal are weighed and their masses are found to be 44.40 g, 40.58 g, and 3...
Questions

History, 13.11.2020 20:10


Mathematics, 13.11.2020 20:10

Mathematics, 13.11.2020 20:10

Mathematics, 13.11.2020 20:10

Mathematics, 13.11.2020 20:10


Mathematics, 13.11.2020 20:10




Mathematics, 13.11.2020 20:10

Mathematics, 13.11.2020 20:10




Chemistry, 13.11.2020 20:10

History, 13.11.2020 20:10

Mathematics, 13.11.2020 20:10

Mathematics, 13.11.2020 20:10

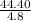 = 9.25g/mL,
= 9.25g/mL, 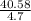 = 8.634g/mL and
= 8.634g/mL and 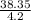 = 9.130g/mL respectively.
= 9.130g/mL respectively.  )÷N}
)÷N}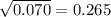 . Thus the standard deviation of the density value is 0.265g/mL.
. Thus the standard deviation of the density value is 0.265g/mL. 

