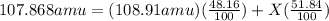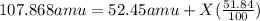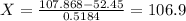Chemistry, 10.07.2019 17:00 lorenzomendi1011
Silver has an atomic mass of 107.868 amu. the ag-109 isotope (108.91 amu) is 48.16%. what is the mass, in amu, of the other isotope? 105.87 amu 106.12 amu 106.91 amu 108.47 amu

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
The drawing represents the movement of particles in a substance. what changes of state can this substance undergo
Answers: 1

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 13:10
What type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? view available hint(s) what type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? salt bridge disulfide bridge hydrogen bond hydrophobic interaction
Answers: 1

Chemistry, 22.06.2019 14:30
Select all of the statements which are true. electrons are located in shells or orbits around the atom. electrons orbit slowly around the atom. electrons travel in one flat path around the nucleus of an atom. the valence of an atom is determined by the number of electrons in the atom's outermost shell.
Answers: 1
You know the right answer?
Silver has an atomic mass of 107.868 amu. the ag-109 isotope (108.91 amu) is 48.16%. what is the mas...
Questions






Mathematics, 25.06.2019 21:00

Mathematics, 25.06.2019 21:00

History, 25.06.2019 21:00

Mathematics, 25.06.2019 21:00

Mathematics, 25.06.2019 21:00

Mathematics, 25.06.2019 21:00

Social Studies, 25.06.2019 21:00


Mathematics, 25.06.2019 21:00




Mathematics, 25.06.2019 21:00

Social Studies, 25.06.2019 21:00

Health, 25.06.2019 21:00