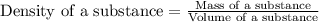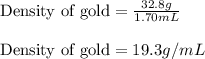Chemistry, 10.07.2019 05:00 pankajyadav7402
A32.8 g sample of gold was submerged in water in a graduated cylinder, and the volume rose by 1.70 ml. what is the density of the gold sample?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
Agroup of students is studying convection current. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other is in an area with warm air. after 10 minutes, the balloon are released from a height of 1 meter. which of the following to the students most likely observe? a) the warm balloon expands and rises. the cold balloon shrinks and sinks b) the balloon both rise. the cold balloon is larger than the warm balloon c) the cold balloon expands and rises. the warm balloon shrinks and sinks d) the balloon rise at the same rate. both balloons are the same size
Answers: 1

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1
You know the right answer?
A32.8 g sample of gold was submerged in water in a graduated cylinder, and the volume rose by 1.70 m...
Questions

History, 20.10.2020 14:01


Mathematics, 20.10.2020 14:01

Social Studies, 20.10.2020 14:01

Mathematics, 20.10.2020 14:01


Mathematics, 20.10.2020 14:01

Mathematics, 20.10.2020 14:01

Law, 20.10.2020 14:01

English, 20.10.2020 14:01

Health, 20.10.2020 14:01




Computers and Technology, 20.10.2020 14:01


Mathematics, 20.10.2020 14:01