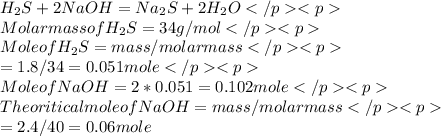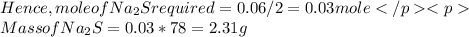Chemistry, 09.07.2019 04:30 papasully1
How many grams of sodium sulfide are formed if 1.80 g of hydrogen sulfide is bubbled into a solution containing 2.40 g of sodium hydroxide, assuming that the sodium sulfide is made in 93.0 % yield?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2


Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 23.06.2019 00:00
What is the approximate mass of 25 cm3 of silver, if the density is 10.5 g/cm3? a. 0.42 g b. 2.4 g c. 42 g d. 260 g
Answers: 1
You know the right answer?
How many grams of sodium sulfide are formed if 1.80 g of hydrogen sulfide is bubbled into a solution...
Questions



Mathematics, 26.01.2021 04:30

Mathematics, 26.01.2021 04:30



Social Studies, 26.01.2021 04:30

Chemistry, 26.01.2021 04:30

Mathematics, 26.01.2021 04:30

Mathematics, 26.01.2021 04:30

Law, 26.01.2021 04:30

Mathematics, 26.01.2021 04:30

Mathematics, 26.01.2021 04:30



Biology, 26.01.2021 04:30

French, 26.01.2021 04:30


Mathematics, 26.01.2021 04:30

Mathematics, 26.01.2021 04:30