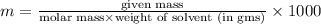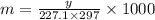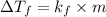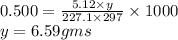Chemistry, 08.07.2019 22:00 adrianty8496
The freezing point of benzene c6h6 is 5.50°c at 1 atmosphere. a nonvolatile, nonelectrolyte that dissolves in benzene is tnt (trinitrotoluene). how many grams of tnt, c7h5n3o6 (227.1 g/mol), must be dissolved in 297.0 grams of benzene to reduce the freezing point by 0.500°c ?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1

You know the right answer?
The freezing point of benzene c6h6 is 5.50°c at 1 atmosphere. a nonvolatile, nonelectrolyte that dis...
Questions

Mathematics, 21.09.2021 14:00



English, 21.09.2021 14:00


Chemistry, 21.09.2021 14:00





Mathematics, 21.09.2021 14:00

Mathematics, 21.09.2021 14:00


History, 21.09.2021 14:00

Mathematics, 21.09.2021 14:00

English, 21.09.2021 14:00




Mathematics, 21.09.2021 14:00

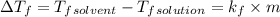
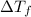 = 0.500°C
= 0.500°C 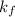 = for benzene is 5.12
= for benzene is 5.12