
Chemistry, 07.07.2019 21:00 christi1175
The decomposition reaction of carbon disulfide to carbon monosulfide and sulfur is first order with k = 2.80 ✕ ✕ 10−7 sec-1 at 1000°c. cs2(g) → cs(g) + s(g) a. how much of a 4.83-gram sample of carbon disulfide would remain after 37.0 days? 1.97 1.97 grams carbon disulfide b. how much carbon monosulfide would be formed after 37.0 days? 1.14 1.65 grams carbon monosulfide useful information 1.013 bar = 760 torr = 1 atm = 760 mm hg

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Amass of 100.0 g of solute is dissolved in water so that 850. ml of a 0.7500 m solution has been prepared. what is the molar mass of the solute?
Answers: 2

Chemistry, 21.06.2019 23:50
2points why do scientists need governmental funding? o a. government politicians ask all the important scientific questions. o b. scientists have to pay taxes to the government on the money they make. o c. the cost of doing scientific research can be very high. o d. the government is controlled by scientists. submit
Answers: 3


Chemistry, 22.06.2019 08:00
An observation that requires measurement is called quantitative observable or qualitative
Answers: 1
You know the right answer?
The decomposition reaction of carbon disulfide to carbon monosulfide and sulfur is first order with...
Questions

Mathematics, 28.01.2021 20:20

Spanish, 28.01.2021 20:20

Spanish, 28.01.2021 20:20


Arts, 28.01.2021 20:20

Computers and Technology, 28.01.2021 20:20


Mathematics, 28.01.2021 20:20


Mathematics, 28.01.2021 20:20

Mathematics, 28.01.2021 20:20




Mathematics, 28.01.2021 20:20

History, 28.01.2021 20:20

History, 28.01.2021 20:20



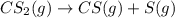
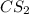 utilised in the reaction
utilised in the reaction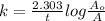
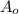 = Initial mass of reactant
= Initial mass of reactant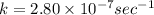
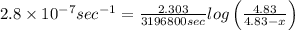
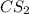 remained after 37 days = 4.83 - x
remained after 37 days = 4.83 - x

