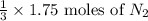Chemistry, 06.07.2019 01:00 biancabahena04
If you have 3.5 mol of n2, and 1.75 mol of h2, how many moles of n2 are there in excess? reaction: n2 + 3 h2 → 2 nh3

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:20
Describing intermolecular forces use the drop down menus to match the type of intermolecular force to its name dipole dipole interactions dipole induced dipole interactions london dispersion forces hydrogen bond van der waals forces done
Answers: 1

Chemistry, 23.06.2019 00:00
#7 how does the structure of amino acids allow them to form a polypeptide? each amino acid has an amino group and a carboxyl group. each amino acid has a hydrogen atom and a carboxyl group. each amino acid has a carboxyl group and an r group. each amino acid has an r group and a hydrogen atom.
Answers: 1

Chemistry, 23.06.2019 00:30
If there are 3.5 moles of koh, how many moles of naoh can be produced? question 1 options: a)3.0 moles naoh b)3.5 moles naoh c)1 moles naoh d)9 moles naoh
Answers: 1

Chemistry, 23.06.2019 08:20
At which temperature would a reaction with ah= -220 kj/mol and as=-0.05 kj/(mol-k) be spontaneous?
Answers: 2
You know the right answer?
If you have 3.5 mol of n2, and 1.75 mol of h2, how many moles of n2 are there in excess? reaction:...
Questions







Mathematics, 04.09.2020 14:01

Spanish, 04.09.2020 14:01


Arts, 04.09.2020 14:01

Social Studies, 04.09.2020 14:01


Chemistry, 04.09.2020 14:01


Mathematics, 04.09.2020 14:01


Physics, 04.09.2020 14:01


Spanish, 04.09.2020 14:01

Mathematics, 04.09.2020 14:01

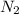 are present in excess.
are present in excess.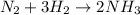
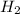 = 1.75 moles
= 1.75 moles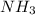 .
.