
Chemistry, 05.07.2019 06:30 yesman1288
In an experiment, potassium chlorate decomposed according to the following chemical equation. kclo3 → kcl + o2 (molar mass of kclo3 = 122.5 g/mol; kcl = 74.55 g/mol; o2 = 31.998 g/mol) if the mass of kcl produced was 180 grams, which of the following calculations can be used to determine the mass of potassium chlorate decomposed?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Read these sentences from the text. near the equator, the tropics receive the most rain on a consistent basis. as a result, the fresh water falling into the ocean decrease the salinity of the surface water in that region. [. .] . . as the salt content of sea water increases, so does its density. what can you infer about how rain affects the density of surface water near the equator?
Answers: 1

Chemistry, 22.06.2019 02:40
Consider the nuclear equation below. 239/94 pu—-> x+ 4/2 he. what is x?
Answers: 2

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

Chemistry, 22.06.2019 10:30
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1
You know the right answer?
In an experiment, potassium chlorate decomposed according to the following chemical equation. kclo3...
Questions

English, 02.04.2020 21:07





History, 02.04.2020 21:07


Chemistry, 02.04.2020 21:07






Spanish, 02.04.2020 21:08



Biology, 02.04.2020 21:08

Mathematics, 02.04.2020 21:08


Geography, 02.04.2020 21:08

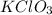 = 122.5 g/mole
= 122.5 g/mole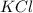 = 74.55 g/mole
= 74.55 g/mole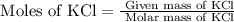
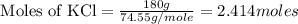
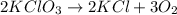
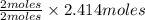 = 2.414 moles of
= 2.414 moles of 

