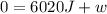Chemistry, 04.07.2019 08:30 dinapaul424
The standard molar heat of fusion of ice is 6020 j/mol. calculate q, w, and ∆e for melting 1.00 mol of ice at 0◦c and 1.00 atm pressure. 1. q = 6020 j/mol

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:30
The reaction q+r2=r2q is found to be first order in r2 and
Answers: 1

Chemistry, 22.06.2019 12:30
Nebulae are enormous clouds in outer space. they are made mostly of hydrogen gas, helium gas, and dust. some nebulae glow brightly, while others do not. the stars that people see are huge, bright balls of glowing gas. they are made mostly of hydrogen and helium. which statement correctly describes other ways in which nebulae and stars are different? a. stars can form inside a nebula but a nebula can never be produced by any star. b. a star always has a higher density than a nebula. c. stars can never form inside a nebula but a nebula can be produced by any star. d. a nebula always has a higher density than a star.
Answers: 3

Chemistry, 22.06.2019 14:20
7. in the cycle, a virus integrates its dna into the host's dna, and its dna is replicated when the host dna is replicated. a. infectious b. retroviral c. lysogenic d.lytic
Answers: 1

Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3
You know the right answer?
The standard molar heat of fusion of ice is 6020 j/mol. calculate q, w, and ∆e for melting 1.00 mol...
Questions



Mathematics, 09.11.2020 16:30


Chemistry, 09.11.2020 16:30



Social Studies, 09.11.2020 16:40





Geography, 09.11.2020 16:40





Engineering, 09.11.2020 16:40

Computers and Technology, 09.11.2020 16:40

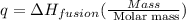 (in terms of mass)
(in terms of mass)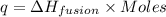 (in terms of moles)
(in terms of moles)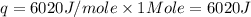
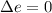
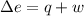
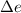 is internal energy.
is internal energy.