
Chemistry, 04.07.2019 07:30 chasereynolds6302
How many formula units of nano3 are in 9.82 moles of nano3 (mw. 85.00 g/mol)?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 05:30
Astudent carefully transfers 30 g of water and 30 g of alcohol in a glass tube, forming two layers and filling the tube completely. after sealing the tube, the student mixes the solutions, and notices a bubble that forms in the tube. what is the mass of the contents in the glass tube after mixing?
Answers: 2

Chemistry, 22.06.2019 15:30
The reactions of photosynthesis occur in the of plant cell? a.mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1
You know the right answer?
How many formula units of nano3 are in 9.82 moles of nano3 (mw. 85.00 g/mol)?...
Questions

Mathematics, 01.10.2019 01:40

Chemistry, 01.10.2019 01:40

Mathematics, 01.10.2019 01:40



World Languages, 01.10.2019 01:40

History, 01.10.2019 01:40



Mathematics, 01.10.2019 01:40

Computers and Technology, 01.10.2019 01:40



Biology, 01.10.2019 01:40

Spanish, 01.10.2019 01:40

Mathematics, 01.10.2019 01:40


History, 01.10.2019 01:40


Health, 01.10.2019 01:40

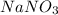 is an ionic compound. So, 1 mol of
is an ionic compound. So, 1 mol of 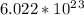 formula units.
formula units.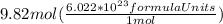
 formula units
formula units

