
Two solutions namely, 500 ml of 0.50 m hcl and 500 ml of 0.50 m naoh at the same temperature of 21.6 are mixed in a constant-pressure calorimeter. the heat capacity of the calorimeter was 450 j/c. given that the specific heat of the solution is 4.184 j/gc, the density of the solution is 1.0 g/ml, and that the heat of neutralization for the process h+oh=h2o is -56.2 kj, what is the final temperature of the mixed solution

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Balance this equation co2(g) + h2o (g) show that the balanced equation obeys the law if conversation of mass
Answers: 1

Chemistry, 22.06.2019 02:00
Which of the following is not a good technique for managing used oil? a) have specific, labeled catch pans available for technicians who are collecting oil b) spills in your shop and any releases on pavement or outside should be poured down a drain c) do not use oil containers for antifreeze or other non-similar fluids d) be prepared for oil spills with the proper absorbents
Answers: 1

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 10:30
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1
You know the right answer?
Two solutions namely, 500 ml of 0.50 m hcl and 500 ml of 0.50 m naoh at the same temperature of 21.6...
Questions


Chemistry, 01.07.2020 15:01


Physics, 01.07.2020 15:01


Mathematics, 01.07.2020 15:01






Physics, 01.07.2020 15:01








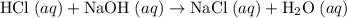
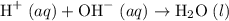
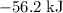 . Thus the combination of every mole of hydrogen ions and hydroxide ions in solution would produce
. Thus the combination of every mole of hydrogen ions and hydroxide ions in solution would produce 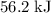 or
or 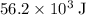 of energy.
of energy.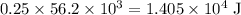 of energy.
of energy. 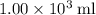 of the 1.0 gram per milliliter solution. Accordingly, it would have a mass of
of the 1.0 gram per milliliter solution. Accordingly, it would have a mass of 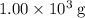 .
. 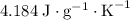 . The solution thus have a heat capacity of
. The solution thus have a heat capacity of 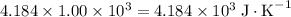 . Note that one degree Kelvins K is equivalent to one degree celsius ℃ in temperature change measurements.
. Note that one degree Kelvins K is equivalent to one degree celsius ℃ in temperature change measurements.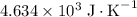 , meaning that its temperature would rise by 1 degree celsius on the absorption of 4.634 × 10³ joules of energy.
, meaning that its temperature would rise by 1 degree celsius on the absorption of 4.634 × 10³ joules of energy.  are available from the reaction. Thus, the temperature of the system shall have risen by 3.03 degrees celsius to 24.6 degrees celsius by the end of the reaction.
are available from the reaction. Thus, the temperature of the system shall have risen by 3.03 degrees celsius to 24.6 degrees celsius by the end of the reaction.

