
Chemistry, 03.07.2019 15:00 aesmithswhs
Alab technician needs to create 570.0 milliliters of a 2.00 m solution of magnesium chloride (mgcl2). to make this solution, how many grams of magnesium chloride does the technician need? refer to the periodic table for . express your answer to three significant figures. the required mass is grams.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:00
A5.000 g sample of niso4 h2o decomposed to give 2.755 g of anhydrous niso4. what is the formula of the hydrate? what is the full chemical name for the hydrate? what is the molar mass of the hydrate? niso4•_h2o what is the mass % of water in the hydrate?
Answers: 1

Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3

Chemistry, 22.06.2019 18:50
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1

Chemistry, 23.06.2019 03:00
What volume does 1.70 ×10–3 mol of chlorine gas occupy if its temperature is 20.2 °c and its pressure is 795 mm hg?
Answers: 3
You know the right answer?
Alab technician needs to create 570.0 milliliters of a 2.00 m solution of magnesium chloride (mgcl2)...
Questions

Mathematics, 15.02.2022 14:00

Mathematics, 15.02.2022 14:00

Physics, 15.02.2022 14:00

English, 15.02.2022 14:00

History, 15.02.2022 14:00

Social Studies, 15.02.2022 14:00


Mathematics, 15.02.2022 14:00



Biology, 15.02.2022 14:00

Mathematics, 15.02.2022 14:00



Mathematics, 15.02.2022 14:00





Business, 15.02.2022 14:00

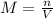 , where M is molarity, n is number of moles and V is the volume. With this equation we can find the number of moles. The volume of 570ml should be converted to L for correct units.
, where M is molarity, n is number of moles and V is the volume. With this equation we can find the number of moles. The volume of 570ml should be converted to L for correct units.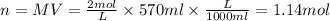
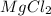 which is 95.21 g/mol to get the mass.
which is 95.21 g/mol to get the mass.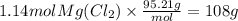 .
.

