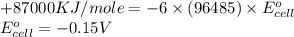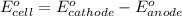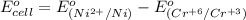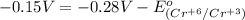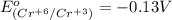3ni2+(aq) + 2 cr(oh)3(s) + 10 oh− (aq) → 3 ni(s) + 2 cro42−(aq) + 8 h2o(l) δg∘ = +87 kj/mol given the standard reduction potential of the half-reaction ni2+(aq) + 2 e− → ni(s) e∘red = -0.28 v, calculate the standard reduction potential of the half-reaction cro42−(aq) + 4 h2o(l) + 3 e− → cr(oh)3(s) + 5 oh−(aq)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Compare and contrast the bohr model and the electron cloud models of the atom.
Answers: 1

Chemistry, 22.06.2019 14:30
An object resting on a table weighs 100 n. with what force is the object pushing on the table? with what force is the table pushing on the object? explain how you got your answer.
Answers: 3

Chemistry, 22.06.2019 19:00
Which is the solubility product expression for caf2(s)?  [ca2+]/[f–]2  [ca2+][f2–]  [ca]+[f]2  [ca2+][f–]2
Answers: 3

Chemistry, 22.06.2019 20:00
The picture represents the process that produces most of the energy used by living organisms on earth. which process is represented in the picture? a) the magnetic attraction between two hydrogen nuclei. b) the fusion of hydrogen nuclei to produce a helium nucleus in the core of the sun. c) the fission of hydrogen nuclei to produce a helium nucleus in the core of the sun. d) the chemical reaction between hydrogen nuclei to produce a helium nucleus in earth's atmosphere.
Answers: 3
You know the right answer?
3ni2+(aq) + 2 cr(oh)3(s) + 10 oh− (aq) → 3 ni(s) + 2 cro42−(aq) + 8 h2o(l) δg∘ = +87 kj/mol given th...
Questions




History, 05.11.2019 18:31




Mathematics, 05.11.2019 18:31

Mathematics, 05.11.2019 18:31


Mathematics, 05.11.2019 18:31

Physics, 05.11.2019 18:31

Advanced Placement (AP), 05.11.2019 18:31

Health, 05.11.2019 18:31

English, 05.11.2019 18:31

Spanish, 05.11.2019 18:31




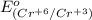 is -0.13 V.
is -0.13 V.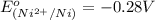
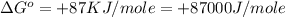 (1 KJ = 1000 J)
(1 KJ = 1000 J)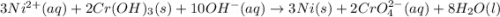
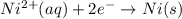
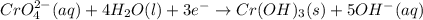
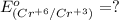
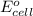 by using formula,
by using formula,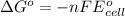
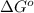 = Gibbs's free energy
= Gibbs's free energy