
Chemistry, 02.07.2019 09:30 alexa006ox9k63
If the density of mercury is 1.36 × 10 by 4 kgm - 3 at 0 degrees. calculate its value at 100 degrees and at 22 degrees. take cubic expansivity of mercury as equal to 180 × 10-6 k - 1

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:00
Which sentence about particles in matter is true? a. atoms are present in solids and liquids but not in gases. b. the particles of matter are in constant motion. c. the same kinds of atoms are found in different elements. d. when a solid changes to a liquid, the sizes of the particles change.
Answers: 1

Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 23.06.2019 02:00
Butane gas reacts with oxygen gas to give carbon dioxide gas and water vapor (gas). if you mix butane and oxygen in the correct stoichiometric ratio, and if the total pressure of the mixture is 390 mmhg, what is the pressure (in mmhg) of water vapor after the reaction is completed (temperature and volume do not change).
Answers: 2
You know the right answer?
If the density of mercury is 1.36 × 10 by 4 kgm - 3 at 0 degrees. calculate its value at 100 degrees...
Questions

Spanish, 31.07.2019 03:30


English, 31.07.2019 03:30

Social Studies, 31.07.2019 03:30

Geography, 31.07.2019 03:30

Mathematics, 31.07.2019 03:30


Mathematics, 31.07.2019 03:30

Mathematics, 31.07.2019 03:30


Geography, 31.07.2019 03:30

Mathematics, 31.07.2019 03:30

Mathematics, 31.07.2019 03:30


Mathematics, 31.07.2019 03:30

Mathematics, 31.07.2019 03:30

History, 31.07.2019 03:30


English, 31.07.2019 03:30

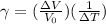 Multiply by V₀ΔT and transpose
Multiply by V₀ΔT and transpose



