
Which of the following statements is true about the strength of the intermolecular forces in ch4 and nh3? a: ch4 ≥ nh3 because ch4 is tetrahedral but nh3 is pyramidal. b: ch4 < nh3 because δ− on c in the ch bond is greater than δ− on n in the nh bond. c: ch4 < nh3 because the nh bond is more polar than the ch bond. d: ch4 ≥ nh3 because ch4 has h bonding but nh3 has dispersion forces.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 07:00
Which atom or ion is the largest? a. k b. k+ c. ca d. ca2+ e. li
Answers: 1


You know the right answer?
Which of the following statements is true about the strength of the intermolecular forces in ch4 and...
Questions

Mathematics, 12.02.2021 17:40


History, 12.02.2021 17:40

Chemistry, 12.02.2021 17:40

Mathematics, 12.02.2021 17:40


Health, 12.02.2021 17:40

Mathematics, 12.02.2021 17:40


Physics, 12.02.2021 17:40


Computers and Technology, 12.02.2021 17:40

Mathematics, 12.02.2021 17:40


Mathematics, 12.02.2021 17:40


Mathematics, 12.02.2021 17:40

Mathematics, 12.02.2021 17:40

Mathematics, 12.02.2021 17:40

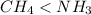 because the NH bond is more polar than CH bond.
because the NH bond is more polar than CH bond.

