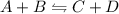Which of the following is not a result when a change to an equilibrium system is applied? increasing the rate of the forward reaction will cause a shift to the left. increasing the rate of the reverse reaction will cause a shift to the left. decreasing the rate of the forward reaction will cause a shift to the left. decreasing the rate of the reverse reaction will cause a shift to the right.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:30
What is i fracture in the crust called when land move up, down or sideways
Answers: 2

Chemistry, 22.06.2019 14:00
How is the atomic number of a nucleus changed by alpha decay
Answers: 2

Chemistry, 22.06.2019 17:00
The msds for glacial acetic acid says that it is a flammable liquid that can severely burn any human tissue it comes in contact with. it reacts with bases, various metals, and strong oxidizing agents. its vapors can form explosive mixtures with air.
Answers: 1

Chemistry, 22.06.2019 20:20
The characteristics of two different types of reactions are shown below: reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of an element. which statement is true about the atoms of the elements that participate in the two reactions? their identity changes in both reaction a and reaction b. their identity changes in reaction a but not in reaction b. their identity changes in reaction b but not in reaction a. their identity remains the same in both reaction a and reaction b.
Answers: 1
You know the right answer?
Which of the following is not a result when a change to an equilibrium system is applied? increasin...
Questions

Arts, 26.11.2020 06:10

Social Studies, 26.11.2020 06:10


Physics, 26.11.2020 06:10

Mathematics, 26.11.2020 06:10


Spanish, 26.11.2020 06:10



Mathematics, 26.11.2020 06:10


History, 26.11.2020 06:10

Business, 26.11.2020 06:10



English, 26.11.2020 06:10

Mathematics, 26.11.2020 06:10

Social Studies, 26.11.2020 06:10