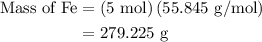Chemistry, 01.07.2019 13:00 amycressey1970
The following data was collected when a reaction was performed experimentally in the laboratory. fe2o3 al al2o3 fe starting amount in reaction 3 moles 5 moles ? ? determine the maximum amount of fe that was produced during the experiment. explain how you determined this amount.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2

Chemistry, 22.06.2019 00:30
Water (2510 g ) is heated until it just begins to boil. if the water absorbs 5.09×105 j of heat in the process, what was the initial temperature of the water?
Answers: 3

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 2
You know the right answer?
The following data was collected when a reaction was performed experimentally in the laboratory. fe2...
Questions


Mathematics, 15.12.2020 14:00

Mathematics, 15.12.2020 14:00

Mathematics, 15.12.2020 14:00

English, 15.12.2020 14:00

Mathematics, 15.12.2020 14:00

Mathematics, 15.12.2020 14:00

Mathematics, 15.12.2020 14:00

Chemistry, 15.12.2020 14:00

Mathematics, 15.12.2020 14:00


Social Studies, 15.12.2020 14:00


Mathematics, 15.12.2020 14:00

Mathematics, 15.12.2020 14:00

Mathematics, 15.12.2020 14:00


English, 15.12.2020 14:00

Mathematics, 15.12.2020 14:00

World Languages, 15.12.2020 14:00

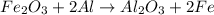
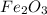 = 3 moles
= 3 moles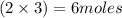 of Al
of Al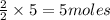 of Fe.
of Fe.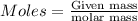
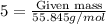
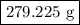 of Fe is produced during the given experiment.
of Fe is produced during the given experiment.
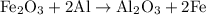
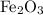 reacts with two moles of Al to produce one mole of
reacts with two moles of Al to produce one mole of 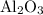 and two moles of Fe.
and two moles of Fe.
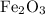 and 5 moles of Al. Therefore the amount of Fe produced by 3 moles of
and 5 moles of Al. Therefore the amount of Fe produced by 3 moles of 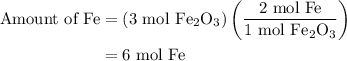
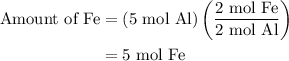
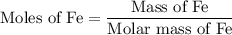 ...... (1)
...... (1) 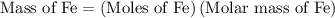 ...... (2)
...... (2)