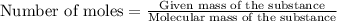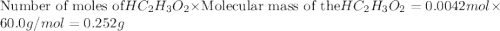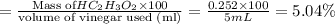The formula to determine the percent of hc2h3o2 (mass/volume) in vinegar is percent (m/v) = (grams of hc2h3o2/ volume of vinegar used (ml) ) x 100. if 5.0 ml of vinegar were used for the titration and 0.0042 moles of hc2h3o2 were required to reach the endpoint, calculate the percent of hc2h3o2 in vinegar. the molar mass of hc2h3o2 is 60.0 g / mol. question 10 options:

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Compare the valence electron configuration of the nobles gas elements seen here. what statement is correct?
Answers: 2

Chemistry, 22.06.2019 16:50
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1

Chemistry, 22.06.2019 19:00
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1

Chemistry, 22.06.2019 22:00
Choose all the answers that apply. fluorine (f) has an atomic number of 9 and an atomic weight of 18.99. fluorine has a. 9 protons b. 10 neutrons c. 18 electrons d. an atomic mass of 19 e. at least one isotope
Answers: 1
You know the right answer?
The formula to determine the percent of hc2h3o2 (mass/volume) in vinegar is percent (m/v) = (grams o...
Questions



Social Studies, 18.11.2019 18:31

Mathematics, 18.11.2019 18:31


History, 18.11.2019 18:31

English, 18.11.2019 18:31


Physics, 18.11.2019 18:31



Mathematics, 18.11.2019 18:31

Mathematics, 18.11.2019 18:31



Social Studies, 18.11.2019 18:31

Mathematics, 18.11.2019 18:31

Mathematics, 18.11.2019 18:31


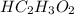 in vinegar 5.04%.
in vinegar 5.04%.