
Chemistry, 01.07.2019 11:00 lisacarter0804
Calculate the mass of 25,000 molecules of nitrogen gas. (1 mole = 6.02 x 1023 molecules)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 12:50
Which phrase best describes the rock's texture? 1.jagged grains 2.coarse grains 3.rounded grains 4.non-banded grains
Answers: 1

Chemistry, 22.06.2019 02:30
Which element forms an ionic bond with flourine? 1) fluorine 2) carbon 3) potassium 4) oxygen
Answers: 1

Chemistry, 22.06.2019 07:20
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1

Chemistry, 22.06.2019 11:30
Voltaic cells produce a positive overall charge. what does this indicate? a. the reaction is likely to be endothermic. b. the reaction is spontaneous. c. the reaction is not likely to occur. d. the reaction is not spontaneous.
Answers: 3
You know the right answer?
Calculate the mass of 25,000 molecules of nitrogen gas. (1 mole = 6.02 x 1023 molecules)...
Questions

Computers and Technology, 04.11.2021 16:30


Health, 04.11.2021 16:30

English, 04.11.2021 16:30


Biology, 04.11.2021 16:30

Biology, 04.11.2021 16:30

Computers and Technology, 04.11.2021 16:30

Mathematics, 04.11.2021 16:30

Mathematics, 04.11.2021 16:30

Mathematics, 04.11.2021 16:30

Mathematics, 04.11.2021 16:30

Chemistry, 04.11.2021 16:40

History, 04.11.2021 16:40

English, 04.11.2021 16:40


Mathematics, 04.11.2021 16:40

Advanced Placement (AP), 04.11.2021 16:40

History, 04.11.2021 16:40

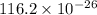 grams
grams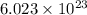 of particles.
of particles.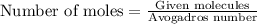
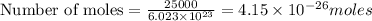
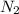 gas weighs = 28 grams
gas weighs = 28 grams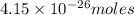 of
of 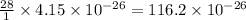 grams
grams

