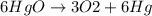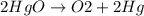Chemistry, 01.07.2019 04:30 Joshua10687
Balance the equation: → + 3hgo → 6o2 + 3hg 3hgo → 3o2 + 3hg 6hgo → 3o2 + 6hg the equation is already balanced

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:40
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 12:10
Building glycogen from glucose molecules is an example of
Answers: 3

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 01:00
Aman applies a force of 500n to push a truck 100m down the street how much does he do?
Answers: 1
You know the right answer?
Balance the equation: → + 3hgo → 6o2 + 3hg 3hgo → 3o2 + 3hg 6hgo → 3o2 + 6hg the equation is alr...
Questions

Mathematics, 21.03.2021 14:00

Computers and Technology, 21.03.2021 14:00

Mathematics, 21.03.2021 14:00


Mathematics, 21.03.2021 14:00


English, 21.03.2021 14:00


Mathematics, 21.03.2021 14:00

Mathematics, 21.03.2021 14:00


English, 21.03.2021 14:00

English, 21.03.2021 14:00

Physics, 21.03.2021 14:00


History, 21.03.2021 14:00